In the realm of chemistry, a fundamental understanding of substances and their reactions begins with the mastery of conversions between molar mass and moles. These quantities provide crucial insights into the proportions and interactions of molecules within chemical equations, opening the door to deciphering the intricate world of chemical reactions.

Image: mungfali.com
Molar mass, expressed in grams per mole (g/mol), represents the mass of one mole of a substance. A mole, on the other hand, is a fundamental unit in chemistry, equivalent to 6.022 × 10^23 particles (atoms, molecules, or ions) of that substance. Understanding the relationship between molar mass and moles is akin to unlocking a treasure chest, revealing the stoichiometry of chemical reactions and the intricacies of chemical equations.
The Golden Key: Converting from Molar Mass to Moles
Just as a treasure map guides explorers to hidden riches, a simple formula serves as the key to converting molar mass to moles:
Moles = Mass (g) / Molar Mass (g/mol)For instance, to determine the number of moles in 25 g of NaCl with a molar mass of 58.44 g/mol:
Moles = 25 g / 58.44 g/mol = 0.428 molesThe Reverse Path: Unraveling Moles into Molar Mass
Unlocking the secrets of molar mass does not end there. Sometimes, the treasure lies in unearthing molar mass from the known number of moles. The formula for this transformation is:
Molar Mass = Mass (g) / MolesUsing the previous example, let’s determine the molar mass of a substance with 0.25 moles and a mass of 14.61 g:
Molar Mass = 14.61 g / 0.25 moles = 58.44 g/mol
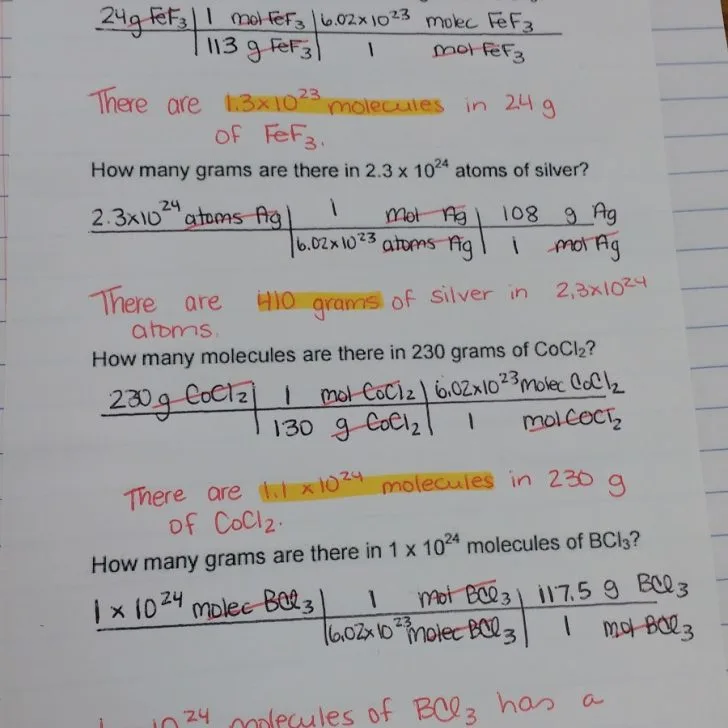
Image: mathequalslove.net
How To Go From Molar Mass To Moles
Realization: Precision and Clarity in Chemical Calculations
Mastering the conversion between molar mass and moles is not merely an academic exercise; it is an essential skill for navig

/GettyImages-1303637-two-way-mirror-57126b585f9b588cc2ed8a7b-5b8ef296c9e77c0050809a9a.jpg?w=740&resize=740,414&ssl=1)



